Radon in Depth: Useful Tips and Specialized Articles
A short history of radioactivity.
What are the different types of radon detectors?
There are several advantages to using our detectors with ionization and electret chambers!
How does a detector with ionization chamber and electret work?
What influences radon infiltration and its concentration?
How is radon harmful and can cause cancer?
Articles

Normes CAN/CGSB-149.11-2024 et CAN/CGSB-149.12-...
Le radon, un gaz radioactif naturellement présent dans le sol, est la deuxième cause de cancer du poumon après le tabac. Invisible, inodore et dangereux lorsqu’il s’accumule dans les bâtiments,...
Normes CAN/CGSB-149.11-2024 et CAN/CGSB-149.12-...
Le radon, un gaz radioactif naturellement présent dans le sol, est la deuxième cause de cancer du poumon après le tabac. Invisible, inodore et dangereux lorsqu’il s’accumule dans les bâtiments,...

Détecteurs de radon : Alpha Track, Corentium Pr...
Le radon est une menace invisible qui peut sérieusement affecter votre santé. En tant que gaz radioactif naturel, il s’infiltre dans les bâtiments et peut provoquer des maladies graves, notamment...
Détecteurs de radon : Alpha Track, Corentium Pr...
Le radon est une menace invisible qui peut sérieusement affecter votre santé. En tant que gaz radioactif naturel, il s’infiltre dans les bâtiments et peut provoquer des maladies graves, notamment...

Entreprise Radon : Spécialiste des Tests et Sol...
Introduction : Protégez Votre Entreprise Contre les Dangers du Radon Le radon, un gaz radioactif invisible, peut s’infiltrer dans les locaux de votre entreprise, représentant un risque sérieux pour vos...
Entreprise Radon : Spécialiste des Tests et Sol...
Introduction : Protégez Votre Entreprise Contre les Dangers du Radon Le radon, un gaz radioactif invisible, peut s’infiltrer dans les locaux de votre entreprise, représentant un risque sérieux pour vos...
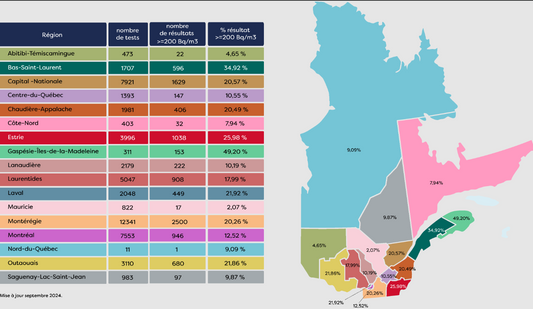
Carte Radon Québec : Votre Guide Essentiel pour...
Le radon, un gaz radioactif naturel, est un enjeu de santé publique au Québec. Invisible et inodore, il s’accumule dans les habitations, augmentant les risques de cancer du poumon. Avec...
Carte Radon Québec : Votre Guide Essentiel pour...
Le radon, un gaz radioactif naturel, est un enjeu de santé publique au Québec. Invisible et inodore, il s’accumule dans les habitations, augmentant les risques de cancer du poumon. Avec...

Pourquoi Tester le Radon dans Votre Maison ?
L'exposition prolongée à des concentrations élevées de radon est la deuxième cause de cancer du poumon après le tabagisme. Étant donné que le radon est indétectable sans équipement spécialisé, il...
Pourquoi Tester le Radon dans Votre Maison ?
L'exposition prolongée à des concentrations élevées de radon est la deuxième cause de cancer du poumon après le tabagisme. Étant donné que le radon est indétectable sans équipement spécialisé, il...

Tout ce que Vous Devez Savoir sur les Tests de ...
Le radon est un gaz inodore et invisible qui peut présenter un risque pour la santé lorsqu'il est présent en concentration élevée à l'intérieur des maisons. Voici un guide complet...
Tout ce que Vous Devez Savoir sur les Tests de ...
Le radon est un gaz inodore et invisible qui peut présenter un risque pour la santé lorsqu'il est présent en concentration élevée à l'intérieur des maisons. Voici un guide complet...
